

Shortages of Medicines in Australia
How to access overseas products in the absence of an Australian registered product
Background
A national medicine shortage occurs when the supply of a medicine is not likely to meet the normal or projected consumer demand within Australia at any point during the next six months. The Therapeutic Goods Administration (TGA) publishes information about national medicine shortages for reportable medicines in the medicine shortage reports database on the TGA website.
There are processes for the supply of medicines that have not been approved for use in Australia by the TGA (i.e. not registered on the Australian Register of Therapeutic Goods – ARTG) that may be accessed at times of medicines shortages. Sometimes these medicines are required for emergency use, and shortages can place patients at risk. It is therefore vital these medicines are available immediately, and there is facility to prescribe ‘unapproved’ medicines from overseas in a clinical setting, as outlined in the case study below. Understandably, part of this process is formal notification to the TGA of ANY use of these vital non-approved medicines.
Case Study: nifedipine immediate release (IR)Nifedipine IR was discontinued in the Australian market. Its ongoing, off-label use in the management of tocolysis (the suppression of premature labour) is critical, particularly in rural and remote regions of Australia. Supplies are needed to be kept in obstetric emergency kits so that it is available when needed. |
Alternative supply options
Prescribers can use the Special Access Scheme (SAS) Category A pathway and the Authorised Prescriber (AP) scheme to access ‘unapproved’ medicines from overseas.
Special Access Scheme (SAS) Category A pathway – Using the medicine
The Special Access Scheme (SAS) allows certain health practitioners to access an ‘unapproved’ therapeutic good for an individual patient on a case-by-case basis. There are three SAS pathways however, the Category A pathway would be most suitable for emergency medicines and does not require the prescriber to wait for approval from the TGA.
The SAS Category A is a notification pathway. Prescribers must submit (online or by email) a completed Category A form to the TGA within 28 days of giving the medicine to the patient. This pathway may be used by a medical practitioner whose patient meets the Category A definition:
Patients who are seriously ill with a condition from which death is reasonably likely to occur within a matter of months, or from which premature death is reasonably likely to occur in the absence of early treatment
How to submit a SAS Category A formThe Category A form is a single page form available on the TGA SAS webpage. It must be completed by a prescribing medical practitioner or by a health practitioner on behalf of a prescribing medical practitioner within 28 days of giving the medicine to the patient and submitted via
|
Holding unapproved goods
The term ‘sponsor’ refers to an individual or company who imports, manufactures and/or supplies medicines in Australia. Unapproved goods must be imported and held under the direct control of the sponsor. For category A SAS medicines, a completed notification form must be submitted to the sponsor so that they may supply the product. It is often necessary to hold emergency supplies on hand for immediate use in Category A patients. Nifedipine for tocolysis is an example of a medicine that is currently prescribed and distributed in this way.
In practice the Australian sponsor and hospitals and clinics may agree on a set of procedures/arrangements to obtain the ‘unapproved’ product prior to the submission of the appropriate forms, including allowing the unapproved product to be held onsite in a controlled location (such as a pharmacy or health centre drug room). The procedures/arrangements between the sponsor and hospital or clinic may include the process of submitting the notification form as well as the process for supply and replacement of stock.
Authorised Prescriber (AP) scheme
An alternate way to obtain emergency medicines in times of shortage is the Authorised Prescriber (AP) scheme. An AP can supply the product directly to patients under their immediate care without requiring separate approval for individual patients. There are 2 pathways to apply to become an AP, depending on the product to be prescribed: The ‘Established history of use pathway’, and the ‘Standard pathway’. The TGA maintains a list of medicines deemed to have an established history of use. As nifedipine IR is not currently on the list of medicines with an established history of use, prescribers can use the standard pathway application process.
How to become an Authorised PrescriberThis requires a 2-step application process:
|
Appendix A
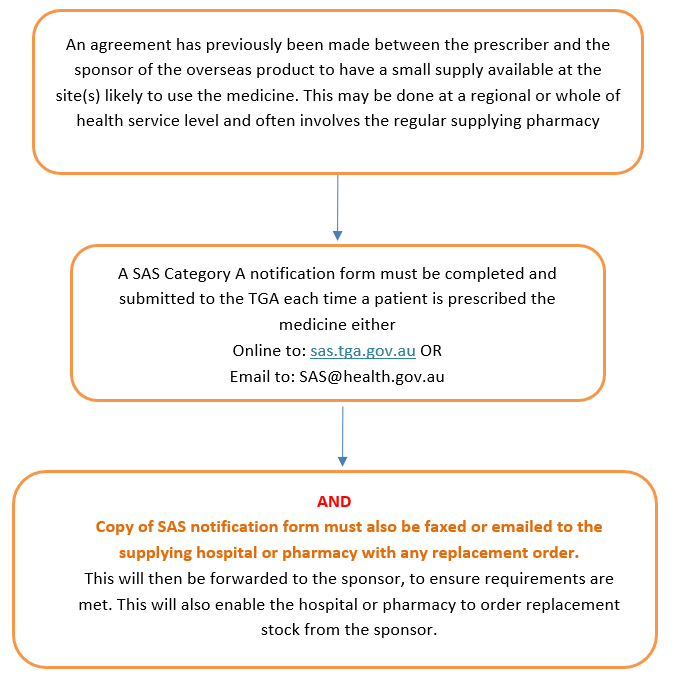
Example flow chart for use of the SAS A pathway
Please refer to the ‘Shortages of Medicines in Australia: How to access overseas products in the absence of an Australian registered product’ document for further information.
 Please find the information provided above here in a printable document.
Please find the information provided above here in a printable document.
Further information
Special Access Scheme (SAS) – https://www.tga.gov.au/form/special-access-scheme
Authorised Prescriber – https://www.tga.gov.au/form/authorised-prescribers
Medicine Shortages Hub – https://www.tga.gov.au/hubs/medicine-shortages

